Table of Contents
Overview
Adverse drug reactions (ADRs) are unintended and harmful effects that occur when medications are used at normal doses. Understanding the types, mechanisms, and risk factors for ADRs is vital for safe prescribing. ADRs can be dose-dependent, immune-mediated, or delayed and may affect any organ system, particularly the liver and kidneys due to their metabolic and excretory roles.
Definition
Adverse drug reactions are any unwanted, harmful effects associated with the use of medications at normal therapeutic doses.
Mechanisms of Adverse Reactions
1. Dose-Related Reactions
- Predictable from the drug’s pharmacological action.
- Often reversible with dose adjustment.
- Examples:
- Anticholinergic side effects (e.g. dry mouth, constipation)
- Sedation or cardiac effects from tricyclic antidepressants
2. Dose-Independent Reactions
- Typically immune-mediated or idiosyncratic.
- Not related to drug concentration.
- Examples:
- Type I hypersensitivity → Allergic reactions (e.g. anaphylaxis, myositis)
- Type II hypersensitivity → Antibody-mediated cytotoxicity (e.g. drug-induced haemolytic anaemia)
Other Types of Adverse Drug Reactions
Carcinogenesis
- Chronic hormone exposure → Hormone-mediated tumour growth
- Cytotoxic drugs → DNA damage and mutation
- Immunosuppressants → Reduce tumour surveillance → viral carcinogenesis
Reproductive Toxicity
- Infertility
- E.g. Alkylating cytotoxic agents (gonadotoxic)
- Teratogenicity (foetal malformations)
- E.g. Anti-epileptics, thalidomide, methotrexate
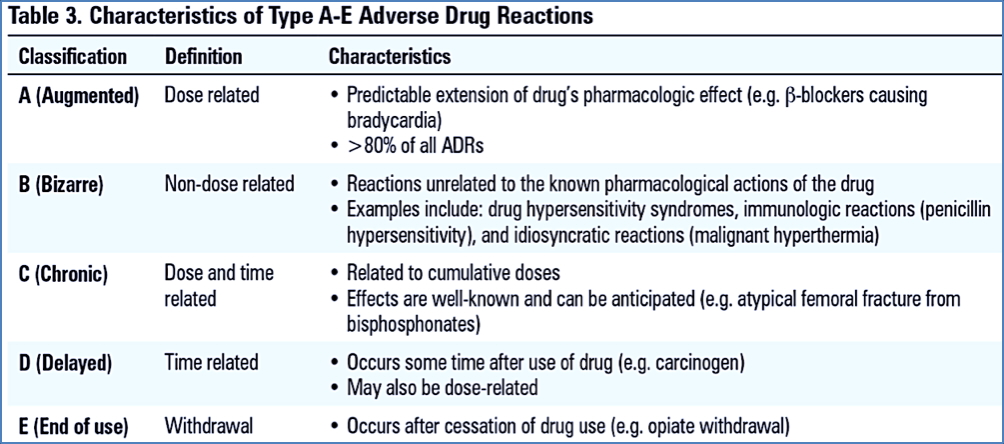
A–E Classification of ADRs

Hepatic and Renal Toxicity
Liver
- Major site of drug metabolism
- Converts drugs into both detoxified and potentially toxic metabolites
- Hepatocytes may be exposed to high levels of toxic intermediates → vulnerable to injury
Kidneys
- Primary organ of drug excretion
- Water-soluble drugs are concentrated in filtrate → Proximal tubule exposed to high doses
- Nephrotoxicity may result from direct tubular exposure
Summary
Adverse drug reactions encompass dose-related toxicity, hypersensitivity responses, and delayed complications like carcinogenesis or teratogenicity. Understanding the A–E classification, immune mechanisms, and organ-specific vulnerabilities helps prevent, detect, and manage drug-related harm. For a broader context, see our Pharmacology & Toxicology Overview page.